Responsibilities of the Principal Investigator
Introduction
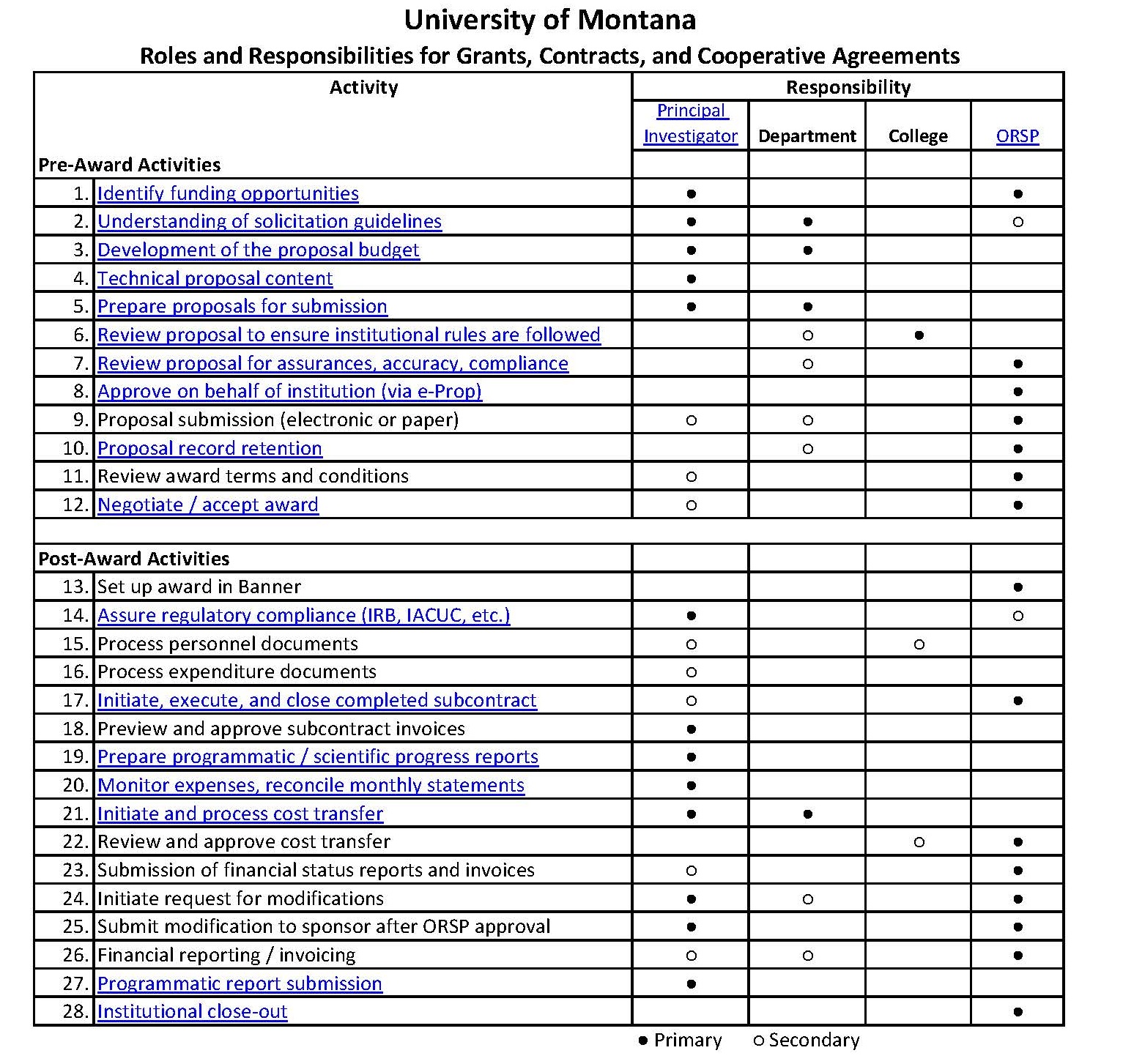
The Project Director (PD) or Principal Investigator (PI) is the primary individual in charge of a grant, contract, cooperative agreement, or other externally sponsored activity. The term "project director" is commonly used in relation to education and public service projects, while the term "principal investigator" is commonly used for research projects. Since the terms are generally used interchangeably by Office of Research and Sponsored Programs (ORSP) and sponsors, we will use the term "PI."
In many cases, there are multiple investigators included in applications for external funding. However, agencies and the University require that one individual be designated as the principal investigator since both the sponsor and the University must have one individual designated as ultimately responsible for the conduct of the project. If co-investigators are named on an application without identifying an individual as the PI, the person listed first is considered the PI by the University and the agency. All PIs and Co-PIs must meet UM's requirements for PI Eligibility.
PI Effort
Leadership and Oversight. Because the PI is responsible for all operation and oversight, a percentage of effort is typically identified and allocated to the activity to reflect the PI's leadership of the sponsored project. Such effort will be allocated to each sponsored project, allocated to the project's related cost-sharing requirements, or paid by other appropriate sources.
Such effort allocation does not apply to equipment and instrumentation grants, dissertation or training grants, or other awards intended as "student augmentation." Similarly, limited purpose grants - such as travel grants or conference support - would involve an insignificant amount of the PI’s time and effort and reflecting a minimum effort threshold is unnecessary.
Proposal Preparation. Time spent on proposal preparation is neither allowable on nor allocable to any existing sponsored activity; a PI should always have a minimum of effort (e.g., 1%) supported by unrestricted funds.
Proposal Preparation and Submission
The Principal Investigator (PI) is responsible for preparing the application for funding to the external sponsor. The PI prepares, or directly supervises the preparation of, both the technical portions of the proposal and the proposal budget. In addition, the PI identifies necessary cost-sharing and/or matching and requests these funds from the appropriate institutional authorities. If the proposed project involves the use of human subjects or laboratory animals, radioactive materials, recombinant DNA/RNA, infectious agents or carcinogens or hazardous materials, the PI must complete, or supervise the completion of, the appropriate compliance documents and obtain the approval of the appropriate University committee.
The PI must also prepare, or directly supervise the preparation of, the University’s proposal checklist form and add the appropriate reviewers as required, typically the department head, dean (or other unit head), and Research and Sponsored Programs. This checklist, with all accompanying documentation, must be institutionally routed and approved prior to the application being submitted to the sponsor.
Signature authority resides with the University, not the PI.
Award Negotiation/Pre-Award Costs
ORSP negotiates any requested changes to the proposed scope of work and budget with the sponsor in collaboration with the PI. If the sponsor requests budget changes, ORSP will work with the PI before submitting a revised budget to the sponsor.
Under the expanded authorities granted to the University by some funding agencies, pre-award expenditures are allowed if prior approval is obtained using the University’s Organizational Prior Approval System (OPAS). The PI prepares or directly supervises the preparation of the OPAS form and must obtain the approval of the department head prior to submitting the completed form to ORSP.
However, PIs should note that such approvals are granted by ORSP with the understanding that the department assumes all risk associated with the establishment of accounts prior to receipt of an award notice from the sponsor. If the award is never received or if the effective date of the award falls outside of the agency-allowed pre-award period, the department will be responsible for all costs incurred.
Management of the Award
The PI is responsible for the proper conduct of the work (performance of the project activities) and for the administrative and financial management of the project.
ORSP staff assist the PI with administrative and financial management of the award. While the University is the legal recipient of most awards and ultimately legally responsible to the sponsor for the performance of the funded activities and the proper use of sponsor funds, the University cannot meet its responsibilities to the sponsor unless the PI meets his/her responsibilities.
Only the PI can truly know if expenses charged to a restricted account are legitimate, project-related costs. As a result, it is the PI's responsibility to regularly (e.g., monthly) review expenses as related to the performance of the project. Similarly, only the PI can assure performance of the technical aspects of the project and completion of sponsor-required project reports.
The University is responsible for maintaining adequate fiscal controls and ensuring that fiscal policies are applied consistently. Therefore, management of externally funded projects is truly a collaboration between the PI and the University.
Project Technical Performance
The PI is responsible for the conduct of project activities in accordance with the sponsor-approved scope of work and in compliance with all University, state, federal, and sponsor policies. This includes hiring, assigning duties to, evaluating, and terminating project staff, scheduling the project activities, completing all sponsor-required technical/progress reports according to the schedule provided by the sponsor, and communicating with the sponsor concerning the conduct of the work and any necessary modifications. The PI is responsible for interacting with any subawardees on the project, and ensuring the programmatic performance of any subrecipients included in the project. ORSP staff can assist the PI if there are difficulties concerning subawardee performance.
Any significant changes to the scope of work and project timetable must be approved by the University and the sponsor. The PI is also responsible for ensuring continued compliance with all University, federal, and sponsor regulations concerning the use of human subjects, laboratory animals, recombinant DNA/RNA, infectious agents, cancer-causing agents, radioactive materials, and hazardous waste for the life of the project. In many cases, this requires the PI to obtain annual approval from the University committees overseeing these activities.
Changes to the PI’s level of effort or that of other key personnel must be approved by the sponsor. The PI is also responsible for complying with University and sponsor requirements concerning the disclosure and protection of project intellectual property.
Project Fiscal Management
The University delegates considerable authority and responsibility for fiscal compliance to principal investigators (PIs) and departments. This requires the development of significant expertise at the department level and relies heavily on PIs and departmental fiscal staff to comply with University policies and various sponsor rules and regulations, and terms and conditions of an award. This includes ensuring that costs are correctly charged to the restricted account according to the sponsor-approved project budget and the terms and conditions of the award.
Each expense charged to the account must directly advance the objectives of the project, must be allowable under the terms and conditions of the award, and must be included in the sponsor-approved budget. Any questions concerning the allowability of costs should be directed to ORSP prior to incurring the cost as ORSP staff can assist the PI in determining if the cost is allowable under the current budget or if a budget revision is required. Note that any expenses incurred within the final 90 days of an award may be closely scrutinized to ensure they fully advance the objectives of the activity.
Departmental administrative support staff assist PIs in managing their accounts, but the PI is ultimately responsible for all costs charged to their restricted accounts. The ORSP post-award staff monitor expenditures against restricted accounts for compliance with University, state, federal, and sponsor guidelines to ensure policies are consistently applied, but only the PI can verify that each individual expense is appropriate to the account.
Ultimately, the PI and the PI’s unit are responsible for any over-expenditures or any costs that are determined to be unallowable by ORSP or auditors. It is therefore incumbent upon the PI to carefully monitor all expenditures charged to his/her restricted accounts and to implement whatever procedures are necessary to ensure compliance with the terms and conditions of the award. To accomplish this, the PI can require that all project-related expenses must be approved in advance by him/her or his/her designee.
-
Personnel Costs
The major expenses in most restricted accounts are personnel costs. To comply with federal regulations, the University has implemented an after-the-fact time and effort reporting system to verify that all personnel costs charged to restricted accounts are appropriate based on the actual time and effort spent on the project. The purpose of the time and effort report is to verify that payroll is distributed appropriately to grant accounts.
If the PI has a number of projects underway, the effort of individual staff members on a specific project may vary from month to month. Because the Principal Investigator is responsible for certifying all time and effort charged to the restricted account, the PI must have a system in place that allows him/her to determine the actual percent effort for each person on each project each month. The University’s time and effort reports must be completed and returned to ORSP on a monthly basis.
To meet his/her responsibilities concerning time and effort reporting, the PI can require each staff person to provide a monthly breakdown of their time and effort on each project. In fact, this is the recommended approach for PIs managing multiple sponsored projects. If the time actually spent on the project differs from the time for which the staff person was paid from the project, the PI must indicate "cost transfer" instead of initialing the incorrect effort before returning it to ORSP. In addition, the PI must prepare and submit, or directly supervise the preparation and submission of, a cost transfer form to ORSP to correct the payroll distribution.
-
Cost Transfers
If the PI determines that costs were charged to the account in error, he/she must prepare, or directly supervise the preparation of, a cost transfer request form. The cost transfer request must specify why the expense is an appropriate cost against the account to which it is being transferred and why it was not charged correctly initially.
According to university policy, all cost transfers must be submitted to ORSP for approval within ninety (90) days of incurring the cost. If ORSP staff see a pattern of inappropriate charges necessitating large numbers of cost transfers, they will contact the PI to discuss modifications to the PI’s procedures to ensure expenses are charged appropriately from the onset.
-
Monitoring Sub-awards
If the project includes subawardees, the PI is responsible for reviewing all invoices submitted by the subawardee and determining that all costs are appropriate and allowable. Payment of subawardee invoices is tied to the subawardee’s performance in meeting the goals outlined in their scope of work. Once the PI has determined the charges are appropriate, the work has been done, and approves the invoice, ORSP staff will process it for payment. The PI must review all such invoices in a timely manner so that the university can meet its responsibility to pay the subawardee in a timely manner.
-
Cost Sharing
It is the PI’s responsibility to document all cost sharing associated with the project. This includes timely submission of time and effort reports, if the cost share is staff time and effort. The PI must provide ORSP with the Banner index number for all University cost sharing committed to the project. If the cost sharing includes contributions from third parties, such as school districts, non-profit agencies, state agencies, and/or private individuals, the PI must obtain written documentation and submit it to ORSP. Cost sharing must be documented as it occurs – the PI should not wait until the project is ending to start documenting cost sharing.
-
Budget Revisions
Because the budget approved by the sponsor was an estimate based on the PI’s knowledge at the time of the application, it is not uncommon for PIs to later determine that the budget needs to be modified to reflect changed circumstances. The PI should contact ORSP as soon as he/she determines modifications of the sponsor-approved budget are needed.
In many cases, sponsors have granted the University the ability to approve some budget changes through the University’s OPAS process. In other cases, a budget revision will need to be approved by the sponsor. All requests to sponsors for budget revisions must be reviewed and approved by ORSP prior to submission to the sponsor.
If you have any questions about your responsibilities or authority as PI, please feel free to contact the Office of Research and Sponsored Programs, (406) 243-6670.